Worksheet - Mendeleev's Periodic Table of Elements Worksheet - Drawing Electron Dot Diagrams Unit Overview & Key Words - Atoms and the Periodic Table Unit PPT - Jeopardy Game: Atoms & the Periodic Table Review UNIT BUNDLE - Atoms and the Periodic Table You Might Also Like the Following Unit Resources: Physical Science - Matter and its Interactions - Structure and Properties of Matter Relevant NGSS Core Idea(s) Addressed by This Product: Download the full PDF preview to see EXACTLY what you are getting.This worksheet comes in 2 formats: a static PDF document and fully-editable WORD document.is a full answer key for Student Review (B) - Basic. 
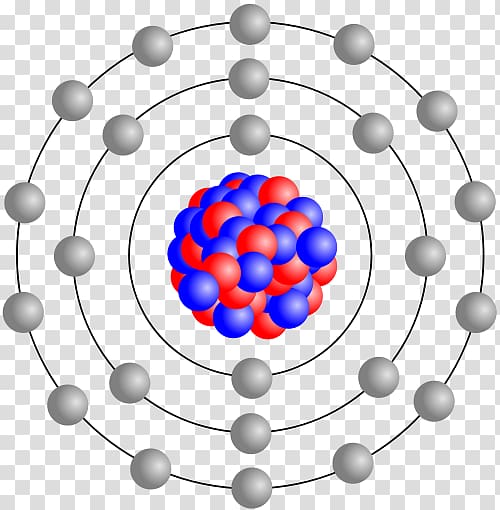
is a full answer key for Student Review (A) - Advanced.is a basic review page that features a fill-in-the-blanks table that closely follows the content of the reading, which allows teachers to provide a more structured exercise for those students that need scaffolding.is an advanced review page that features a blank table that follows the format of the reading and allows students to write their own summary notes in paragraph of point form.Erwin Schrodinger and the quantum model.Ernest Rutherford and the nuclear model.John Dalton and the first atomic theory.Democritus and the idea of atoms (atomos).A 2-page reading that covers the following topics:.Pages 1-2 - History of the Atomic Model Reading: Worksheet - History of the Atomic Model.There is a review quiz that is designed to assess student understanding of the content of this reading.After showing the relevant PPT presentation slides to my students and viewing a brief video clip, I hand out this product for them to work on for the remainder of the lesson and for homework.I typically use this as a summary exercise designed to be a supporting document to my PPT on the atomic model.Review (B) is a more structured fill-in-the-blanks summary.Review (A) gives students more freedom to write their own summary notes in a blank space.This product features 2 differentiated student review pages, which gives educators a choice for which review page best fit the needs of their own class.Thomson and the plum pudding model, Ernest Rutherford and the nuclear model, Niels Bohr and the planetary model, Erwin Schrodinger and the quantum model, and J ames Chadwick and the neutron. A 2-page reading section covers the following topics: what is a scientific model, Democritus and atomos, John Dalton and his atomic theory, J.J. Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics.This 4-page worksheet product is designed to provide a condensed summary of the history of the atomic model for middle school and early high school chemistry students. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
